Though many different types of metals may be etched, copper is one of the most common materials used in both traditional and modern etching contexts. Read More…
Great Lakes Engineering is a trend setting manufacturer of surface mount stencils, precision laser cut parts, and photo chemical etched parts. We work with a wide range of materials, including Stainless Steel, Copper, Titanium, Nitinol, Nickel, Kovar and many others.

VACCO is the industry leader of Photo Chemical Etching of metal & polyimide components and devices. We specialize in Stainless steel, Titanium, and Copper, but work with a variety of different materials. We have over 60 years of experience in Chem Etching, and we offer Micro Laser Cutting & Welding, and Diffusion & Adhesive Bonding services along with an extensive range of value-added services. ...

The MET Manufacturing Group, LLC process offers many technical & financial advantages in manufacturing various flat metal components. Try this precision etching, non-mechanical process for competitively priced, burr & stress free sheet metal products, up to 62 mil (.062”) thick. Our photo-chemical machining process is also known as photo-fabrication, photo etching, chemical milling & acid...
Etchit is your high-quality solution for custom-manufactured precision metal parts and components. We use photochemical machining to make products for such industries as aerospace, audio, automotive, computer, circuit board, decorative and fastener. Does your product need photo etching processes?

Photochemical machining is the process we use on aluminum (and alloys), brass, copper, inconels, kovar, metal foils (less than .001” thick), metal sheets (up to .125” thick), mild steels, molybdenum, monels, nickel, phosphor bronze, stainless (300 & 400) and others. Trust your metal etching needs to us.
More Copper Etching Companies
One of the few metals found abundantly in nature, copper is easily mined, refined, and 100% recyclable. This malleable and ductile metal is easily manufactured into strips, smooth rods or tubes, sheets and plates desirable for engraving.
The fine qualities of copper make it the preferred choice for etching as it bites evenly, holds texture well and color is not often distorted. Although green patina may develop over time when copper is exposed to environmental conditions, artistic etchings often utilize this attribute for a more integrated design.
While hobbyists and artists frequently utilize copper etching, industrial and commercial manufacturing also incorporate this medium in a variety of applications. Aerospace, automotive, transportation, electronics, minting and medical industries all utilize copper etching in metal components.
The popularity of copper in many of these settings can be attributed to its being one of the best known and most cost effective conductors of heat and electricity.
For this reason and others etched copper products include microchips, statues, decorative ornaments, printed circuit boards, musical instruments, piping, tubing, cookware, coinage and even ammunition.
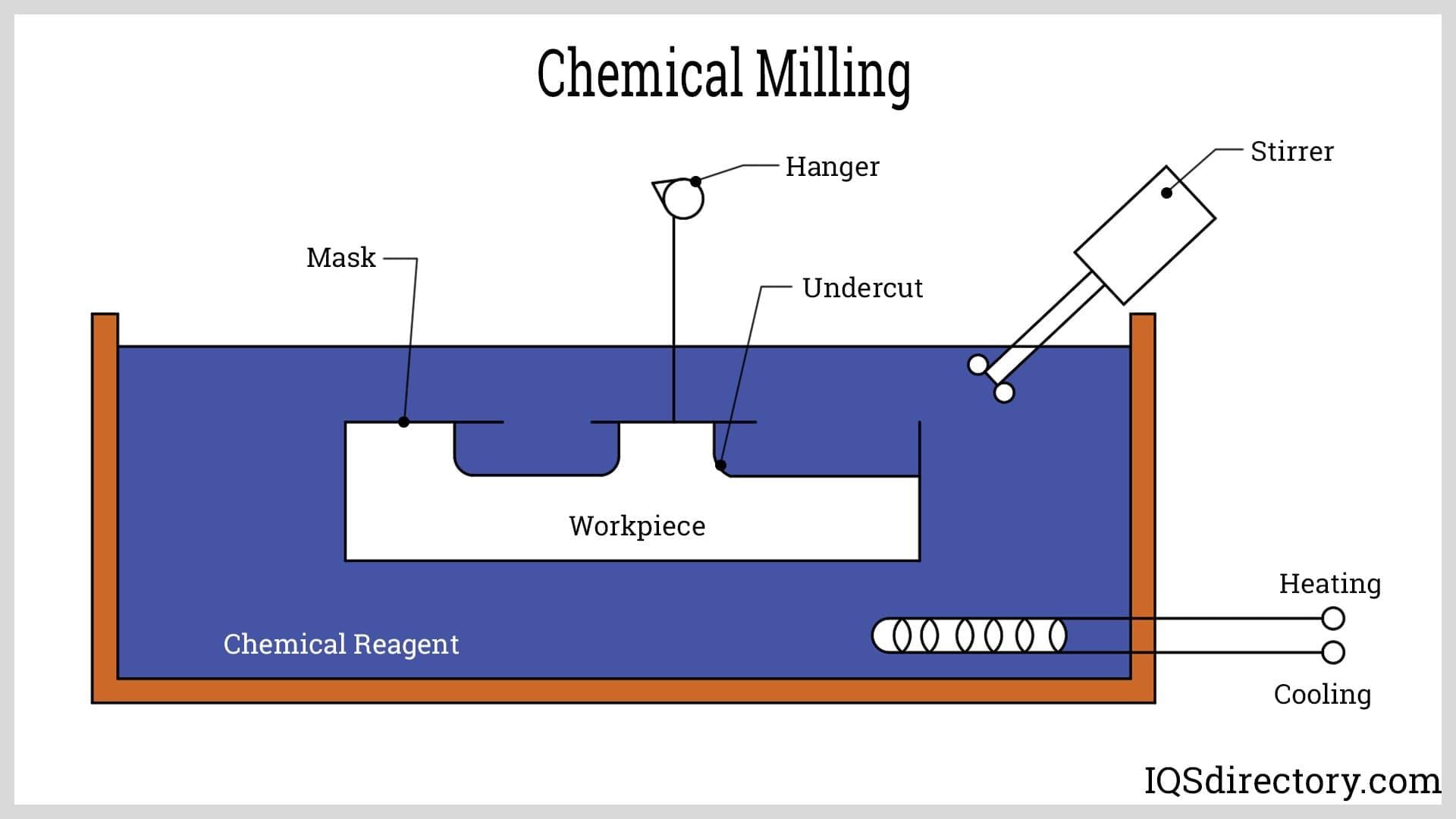
Such a wide variety of copper etched products reflects the many different etching techniques available. While hand etching with a needle is popular in artistic and custom applications, chemical etching or machining is the most common industrial etching method, particularly acid etching and photofabrication.
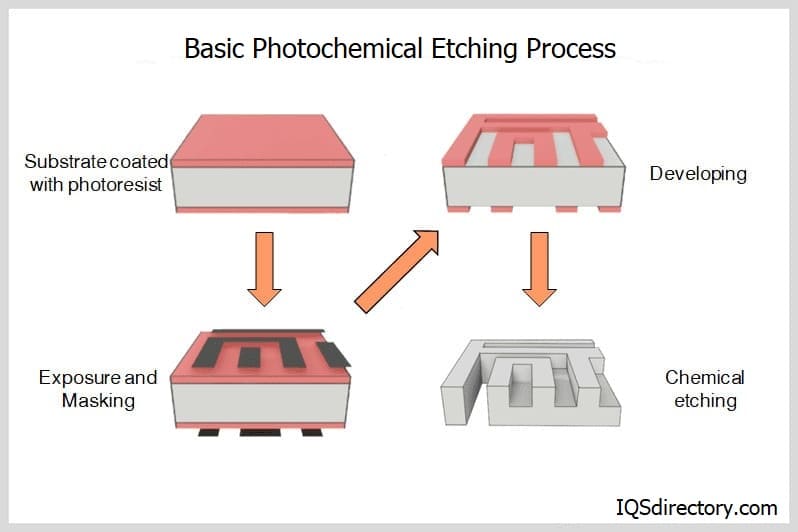
Acid etching is accomplished by covering the copper surface to be etched with an acid resistant waxy ground. The manufacturer scratches off the ground, using a point etching needle creating the design of the finished piece.
The copper sheet is then dipped into acid or has acid washed over the metal. Photofabrication follows a similar procedure but rather than a waxy ground, the copper is covered in a photoresist and exposed to UV light to create the template necessary before the corrosive chemical is applied.
In either instance, the chemical wash bites into the metal where it is unprotected, leaving a depression in the plate. The depth of the groove depends on the length of application.
When the desired depth is achieved, the remaining chemicals are washed off and the rest of the wax or photoresist is removed to reveal the finished product. Cupric chloride is the chemical most often used with copper due to its high etch rate, though iron chloride and nitric acid are also compatible with copper.









 Broaching
Broaching CNC Machining
CNC Machining Expanded Metals
Expanded Metals Laser Cutting
Laser Cutting Metal Etching
Metal Etching Metal Fabrication
Metal Fabrication Perforated Metals
Perforated Metals Screw Machine Products
Screw Machine Products Metal Stampings
Metal Stampings Sheet Metal Fabrication
Sheet Metal Fabrication Tube Fabrication
Tube Fabrication Water Jet Cutting
Water Jet Cutting Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services